CISN - Cancer Research Advocacy - page 3
| You Are Here: Home > Advocacy > Types of Advocacy > Research Advocacy - page 3 | |
AdvocacyAlso in this section: |
|
Research Advocacy - page 3Types of Research Advocacy - continued...Advocates in CLINICAL RESEARCHClinical research is research that either directly involves a particular person or group of people, or studies information about them, such as their behavior. The goal of clinical research in cancer is to develop more effective strategies for cancer prevention, diagnosis and treatment. Clinical research can be divided into the following categories: Patient-oriented research (clinical trials); Epidemiological and behavioral studies. Using our arrow image, you can see we are now at the primary intersection with human patients. All of the work completed in basic and translational research is tested here.
Clinical research includes the conduct of large clinical trials in people. Several different groups conduct clinical trials.
|
| Types of Advocacy | |
| Community Advocacy | |
| ● | Research Advocacy |
| Articles | |
| Community Projects | |
| Collaborations | |
| Calendar | |
 |
|
|---|---|
| http://www.cancer.gov/clinicaltrials/nctn |
What do advocates do in CLINICAL RESEARCH?
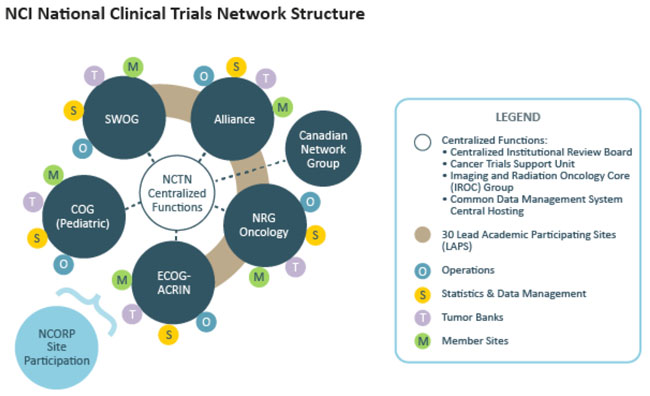
Clinical research involves patients, so the inclusion of advocates and patients is very important. Most researchers, institutions and funders agree on many aspects of advocate involvement in clinical research. The government run National Clinical Trials Network is one area in which advocate involvement is readily seen.
Patient advocates are involved in all aspects of clinical trial development through the National Clinical Trials Network (supported by grants from the National Cancer Institute; see information box) to provide the patients perspective throughout the development and implementation of clinical trials by:
- Initiating strategies that accelerate study development, activation, accrual participation and reporting results
- Aiding in the selection of cancer research concepts to study
- Fostering collaboration and interactions between and among committees
- Ensuring that clinical trials are both patient centered (lead to improved clinical outcomes) and respect patients
Patient advocates can also be involved locally at Institutions in their area (e.g. NCI designated Comprehensive Cancer Centers, Community Cancer Centers). Locally there are many ways that advocates can help to make sure trials accrue, there are no barriers to patient participation and that patients are fully informed before they consent to be on a clinical trial.
Below are a few activities of advocates in clinical research at local cancer centers.
- Every Comprehensive Cancer Center should have a Protocol Committee that reviews every study before it is sent to the Institutional Review Board for final approval.
 |
The advocate role on this committee can vary, but may include ensuring readability and accuracy of the consent form and the identification of potential patient barriers to accrual. |
|---|
- Every institution that conducts research in people (clinical trials) has an Institutional Review Board (IRB). This board reviews every clinical trial to assure that appropriate steps are taken to protect the rights and welfare of people participating in clinical trials (FDA web-site). There is always a consumer advocate on the IRB at any institution conducting clinical trials.
- Patients can be members of local Data Safety Monitoring Boards (DSMB)
- DSMBs are assembled for most clinical trials to provide oversight and monitoring of the conduct of a clinical trial to ensure the safety of participants while maintaining integrity of the data. They are an independent group that can look at the data for undue toxicity or futility (an arm is clearly inferior), without compromising the data. Advocates can participate as voting members of these boards.
- Advocates can become involved with their health care providers in many ways. One way advocates become involved is through navigation of other patients through the system. Many advocates also facilitate support groups and maintain a high level of provider and patient interactions.
Why is it important?
It is very important to include advocates in clinical research projects to ensure the patient point of view is heard during the conduct of this type of research.
 |
Advocates also understand the shock and fear patients experience after a cancer diagnosis and how difficult it is to make treatment decisions. They can help to develop strategies to assist a patients understanding of clinical trials. |
|---|
Additional important contributions are listed below:
- Inclusion of advocates in clinical research decisions fosters public trust.
- Advocates can see barriers to accrual through a patients viewpoint and ensure that they are removed or minimized.
- Advocates help to communicate information about clinical trials by:
- Engaging the broader advocacy community and public
- Developing online resources to educate the public on the availability of clinical trials as an investigational (still be studied) treatment option
- Developing patient centered materials and encouraging physicians to discuss trials with patients
- Publishing and disseminating clinical trial results in plain language to the general public
Advocates in Drug Development
The Food and Drug Administration (FDA) is an agency within the US Department of Health and Human Services and has many responsibilities including protecting the public health by assuring the safety, effectiveness, quality and security of human and animal drugs, vaccines, biological products and medical devices.
Part of the FDAs mission is to evaluate new therapies and determine which are safe and effective for their intended use. This is a complex job, often involving many areas of expertise, and sometimes the FDA turns to outside experts for counsel.
FDAs advisory committees provide independent, expert advice to the agency on a range of complex scientific, technical, and policy issues.
What do advocates do in the FDA?
The FDA has involved advocates in many different ways. The FDA has many different types of meetings and involves patients and consumers during these meetings.
- Some of the meetings are early in drug development and include meetings with the sponsor about how best to move a drug into the approval process.
- Some meetings are during the drug approval process to monitor ongoings
- Others are to evaluate different ways of approving drugs or changes to the approval process.
To include research advocates the FDA recruits Patient Representatives (who are Special Government Employees) to participate as consumer and patient advocates on many different kinds of FDA committees.
Why is it important?
One very important role for advocates at the FDA is when the data is submitted for the approval of an agent but the results are not clearly positive or negative. At this point they may initiate an Oncologic Drugs Advisory Committee (ODAC) meeting.
This committee includes top scientists in the field as well as a consumer representative and a patient representative. Advocates who are survivors can participate as either. Those who are not survivors would only be able to be a consumer representative. The perspective of the patient and consumer is not to be contradictory, but is to evaluate the evidence and ensure the correct decision is made on behalf of all patients that may benefit from the new agent.
 |
This includes an evaluation of the risks and benefits as presented at the meeting. For more information on risk and benefit see important advocate issues in drug development on page 4 of Research Advocacy. |
It is extremely important for the patient and consumer representatives to be well informed and have the ability to understand the disease being reviewed and how it is currently treated.
Below are some benefits patients and consumers bring to an FDA meeting:
- Representatives for industry, patients, and consumers add different perspectives and expertise that give balance to the discussions and final recommendations.
- Patient and consumer representatives facilitate dialogs that affect consumers.
- Patients can evaluate the safety and effectiveness of products that are under review, so they need an ability to understand research and scientific data.
- Evaluating risk versus benefit from a patient perspective is important (see section on risk/benefit).
| Research Advocacy | |||

