CISN - Drug Development
| You Are Here: Home > Cancer Research > Drug Development > CISN Summary of Drug Development Challenges |
|||||||||
CISN Summary of |
 |
Examination of the various parts of the process helps each of the stakeholders to better understand the other parts and to work as cooperatively as possible. |
|
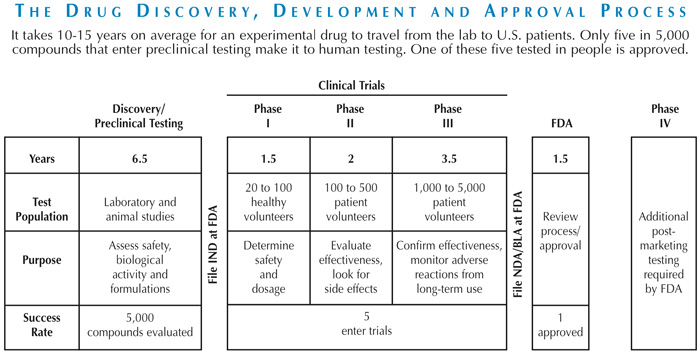
The major challenges of cancer drug development:
- Length of time from early research to drug approval can be up to 20 years on average
- The overall success rate for oncology products in clinical development is only about 10%
- Cost to bring a new drug to market can reach approximately $1 billion
- Resources are often invested in the development of compounds that ultimately are not viable treatments
- Needs of multiple stakeholders are varied and sometimes conflicting
- Multiple clinical trials must be conducted and many patient participants are needed for drug approval
| Section Index | |
| What We Know About Cancer | |
| How Cancer is Studied | |
| ● | Drug Development |
| New Treatments | |
| Research Advocacy | |
You have reached the end of this section. If you wish to test your knowledge of "Drug Development"For more information about our e-Training courses, please read the e-Quiz Introduction. |
| |